Dlí Boyle
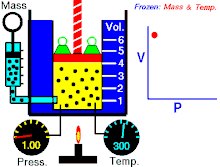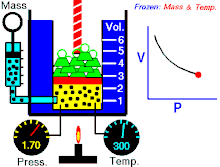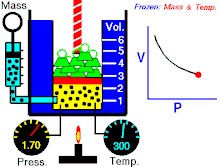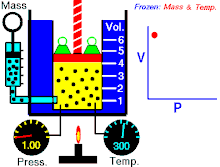
Tá Dlí Boyle nó Dlí Mariotte, dá ngairtear Dlí Boyle-Mariott go minic, a ainmníodh i ndiaidh an fealsamh nádúrtha agus scríbhneoir diagachta Angla-Éireannach, Robert Boyle agus an luibheolaí agus fisiceoir Francach Edme Mariotte, ar cheann de na dlíthe teirmidinimice arb é dlí an gháis idéalaigh iad. Baineann sé brú agus toirt an gháis idéalach ag teocht tairiseach. Tugann an Dlí seo le fios an méid seo a leanas; "Tá toirt maise gháis i gcomhreir inbhéartach leis an mbrú ar an ngás ag teocht sheasmhach."
Nuair a mhéadaítear an brú laghdaítear an toirt, i bhfocail eile, i bhfoirm chothromóide
| Tá an brú i gcomhréir inbhéirteach leis an toirt | |
nó
| Is ionann brú iolraithe faoina thoirt agus an tairiseach |
Stair
[cuir in eagar | athraigh foinse]
| V (in2) | p (in Hg) |
|---|---|
| 117.5 | 12 |
| 87.2 | 16 |
| 70/7 | 20 |
| 58.8 | 24 |
| 44.2 | 32 |
| 35.3 | 40 |
| 29.1 | 48 |
Thug Richard Towneley agus Henry Power an gaol seo idir brú agus toirt faoi deara den chéad uair sa 17ú haois.[2][3] Dheimhnigh Robert Boyle a bhfionnachtain trí thurgnaimh agus d’fhoilsigh sé na torthaí.[4] Dar le Robert Gunther agus údaráis eile, ba é cúntóir Boyle, Robert Hooke, a thóg an gaireas turgnamhach. Tá dlí Boyle bunaithe ar thurgnaimh le haer, a mheas sé a bheith ina sreabhán cáithníní ar fos idir spriongaí beaga dofheicthe. Ag an am sin, bhreathnaítí fós go raibh aer mar cheann de na ceithre dúil, ach d'easaontaigh Boyle. Is dócha gurbh é a mheas Boyle ná go raibh an t-aer ina ghné riachtanach den saol;[5] mar shampla, d’fhoilsigh sé saothair ar fhás plandaí gan aer.[6] D'úsáid Boyle feadán dúnta ar chruth J agus tar éis dó mearcair a dhoirteadh ó thaobh amháin chuir sé iallach ar an aer ar an taobh eile crapadh faoi bhrú mearcair. Tar éis an turgnamh a athdhéanamh arís agus arís eile agus méideanna éagsúla mearcair a úsáid, bhí brú an gháis i gcomhréir inbhéirteach lena toirt.[7] D’aimsigh an fisiceoir Francach Edme Mariotte (1620–1684) an dlí céanna go neamhspleách ar Boyle i 1679,[8] ach d’fhoilsigh Boyle é cheana féin i 1662.[7] Mariotte did, however, discover that air volume changes with temperature.[9] Fuair Mariotte amach, áfach, go n-athraíonn toirt an aeir le teocht. Uaireanta tugtar dlí Mariotte air seo nó dlí Boyle-Mariotte. Níos déanaí, sa bhliain 1687 sa Philosophiæ Naturalis Principia Mathematica, léirigh Newton go matamaiticiúil go bhfuil an sreabhán leaisteach ina bhfuil cáithníní faoi shuan, agus fórsaí eatru atá i gcomhréir inbhéartach lena n-achar, bheadh an dlús i gcomhréir dhíreach leis an mbrú.[10] In ionad teoiric statach, teastaíonn teoiric chinéiteach, a chuir Maxwell agus Boltzmann ar fáil dhá chéad bliain ina dhiaidh sin.
Is é seo an chéad dlí fisiceach a thuairiscítear i bhfoirm cothromóide ag cur síos ar spleáchas dhá chainníocht athraitheacha.[7]
Sainmhíniú
[cuir in eagar | athraigh foinse]Is féidir an dlí féin a lua mar seo a leanas:
- Maidir le mais sheasta de ghás idéalach a choimeádtar ag teocht sheasta, tá brú agus toirt i gcomhréir inbhéartach.
Nó is dlí gáis é dlí Boyle, á rá go bhfuil gaol inbhéartach ag brú agus toirt gáis. Má mhéadaíonn an toirt, ansin laghdaíonn an brú agus a mhalairt, nuair a choinnítear an teocht seasmhach.
Dá bhrí sin, nuair a dhéantar an toirt a laghdú go leath, déantar an brú a dhúbailt; agus má dhéantar an toirt a dhúbailt, déantar an brú a laghdú go leath.
An gaol le teoiric chinéiteach na ngás idéalach
[cuir in eagar | athraigh foinse]Luann dlí Boyle go bhfuil ag teocht sheasmhach toirt maise áirithe de ghás tirim i gcomhréir inbhéartach leis an mbrú.
Iompraíonn mórchuid na ngás mar [gás idéalach|gáis idéalacha]] ag brúnna agus teochtaí measartha. Ní fhéadfadh teicneolaíocht an 17ú haois teocht an-ard nó an-íseal a tháirgeadh. Dá réir sin, ní dócha go mbeidh claontaí ag an dlí tráth a fhoilsithe. Le himeacht ama, cheadaigh feabhsuithe sa teicneolaíocht. brú níos airde agus teochtaí níos ísle a bhaint amach, agus thugtaí dialltaí ó iompar idéalach na ngás faoi deara, agus ní féidir leat cur síos cruinn a dhéanamh air ach trí theoiric na bhfíorghas a úsáid.[11] Cuirtear síos ar an ndiall mar fachtóir in-chomhbhrúiteachta.
Féach freisin
[cuir in eagar | athraigh foinse]Tagairtí
[cuir in eagar | athraigh foinse]- ↑ "Feadán-J" Boyle. https://javalab.org/en/boyles_j_tube_en/
- ↑ See:
- Henry Power, Experimental Philosophy, in Three Books … (London: Printed by T. Roycroft for John Martin and James Allestry, 1663), pp. 126–130. Ar fáil ar líne: Early English Books Online. On page 130, Power presents (not very clearly) the relation between the pressure and the volume of a given quantity of air: "That the measure of the Mercurial Standard, and Mercurial Complement, are measured onely by their perpendicular heights, over the Surface of the restagnant Quicksilver in the Vessel: But Ayr, the Ayr's Dilatation, and Ayr Dilated, by the Spaces they fill. So that here is now four Proportionals, and by any three given, you may strike out the fourth, by Conversion, Transposition, and Division of them. So that by these Analogies you may prognosticate the effects, which follow in all Mercurial Experiments, and predemonstrate them, by calculation, before the senses give an Experimental [eviction] thereof." In other words, if one knows the volume V1 ("Ayr") of a given quantity of air at the pressure p1 ("Mercurial standard", i.e., atmospheric pressure at a low altitude), then one can predict the volume V2 ("Ayr dilated") of the same quantity of air at the pressure p2 ("Mercurial complement", i.e., atmospheric pressure at a higher altitude) by means of a proportion (because p1 V1 = p2 V2).
- Charles Webster (1965). "The discovery of Boyle's law, and the concept of the elasticity of air in seventeenth century," Archive for the History of Exact Sciences, 2 (6) : 441–502; see especially pp. 473–477.
- Charles Webster (1963). "Richard Towneley and Boyle's Law," Nature, 197 (4864) : 226–228.
- Robert Boyle acknowledged his debts to Towneley and Power in: R. Boyle, A Defence of the Doctrine Touching the Spring and Weight of the Air, … (London, England: Thomas Robinson, 1662). Ar fáil ar líne: Spain's La Biblioteca Virtual de Patrimonio Bibliográfico. On pages 50, 55–56, and 64, Boyle cited experiments by Towneley and Power showing that air expands as the ambient pressure decreases. On p. 63, Boyle acknowledged Towneley's help in interpreting Boyle's data from experiments relating the pressure to the volume of a quantity of air. (Also, on p. 64, Boyle acknowledged that Lord Brouncker had also investigated the same subject.)
- ↑ Gerald James Holton (2001). "Physics, the Human Adventure: From Copernicus to Einstein and Beyond": 270–. Rutgers University Press.
- ↑ R. Boyle, A Defence of the Doctrine Touching the Spring and Weight of the Air, … (London: Thomas Robinson, 1662). Ar fáil ar líne: Spain's La Biblioteca Virtual de Patrimonio Bibliográfico. Boyle presents his law in "Chap. V. Two new experiments touching the measure of the force of the spring of air compress'd and dilated.", pp. 57–68. On p. 59, Boyle concludes that " … the same air being brought to a degree of density about twice as that it had before, obtains a spring twice as strong as formerly." That is, doubling the density of a quantity of air doubles its pressure. Since air's density is proportional to its pressure, then for a fixed quantity of air, the product of its pressure and its volume is constant. On page 60, he presents his data on the compression of air: "A Table of the Condensation of the Air." The legend (p. 60) accompanying the table states: "E. What the pressure should be according to the Hypothesis, that supposes the pressures and expansions to be in reciprocal relation." On p. 64, Boyle presents his data on the expansion of air: "A Table of the Rarefaction of the Air."
- ↑ The Boyle Papers BP 9, fol. 75v–76r at BBK.ac.uk Curtha i gcartlann 2009-11-22 ar an Wayback Machine
- ↑ The Boyle Papers, BP 10, fol. 138v–139r at BBK.ac.uk Curtha i gcartlann 2009-11-22 ar an Wayback Machine
- ↑ 7.0 7.1 7.2 "Scientists and Inventors of the Renaissance" (2012): 94–96. Britannica Educational Publishing.
- ↑ See:
- Mariotte, Essais de Physique, ou mémoires pour servir à la science des choses naturelles, … (Paris, France: E. Michallet, 1679); "Second essai. De la nature de l'air".
- (Mariotte, Edmé), Oeuvres de Mr. Mariotte, de l'Académie royale des sciences; … , vol. 1 (Leiden, Netherlands: P. Vander Aa, 1717); see especially pp. 151–153.
- Mariotte's essay "De la nature de l'air" was reviewed by the French Royal Academy of Sciences in 1679. See: (Anon.) (1733) "Sur la nature de l'air," Histoire de l'Académie Royale des Sciences, 1 : 270–278.
- Mariotte's essay "De la nature de l'air" was also reviewed in the Journal des Sçavans (later: Journal des Savants) on 20 November 1679. See: (Anon.) (20 November 1679) "Essais de physique, … ," Journal des Sçavans, pp. 265–269.
- ↑ Ley, Willy (June 1966). "The Re-Designed Solar System". For Your Information. Galaxy Science Fiction. pp. 94–106.
- ↑ Principia, Sec. V, prop. XXI, Theorem XVI
- ↑ Ira. N. (1978), lch. Tugann 11 dá aire go dtarlaíonn dialltaí le brúnna agus teochtaí arda.



